
The Medtronic InterStim™ System
One of the most exciting therapies our office offers for the treatment of OAB is the InterStim™ system by Medtronic, one of the world’s leading healthcare innovators.
For more than 20 years, InterStim™ has been used to improve bladder and bowel function in more than 250,000 patients. One medical study found that 85% of patients who’ve had the system in place for a year saw significant improvement in their condition2,*. After 5 years, that rate was 82%, and 45% of patients with leaking had no leakage at all3,†. Device-related adverse events occurred in 30% of subjects post-implant. The most common device-related adverse effects were undesirable change in stimulation, implant site pain, and implant site infection.
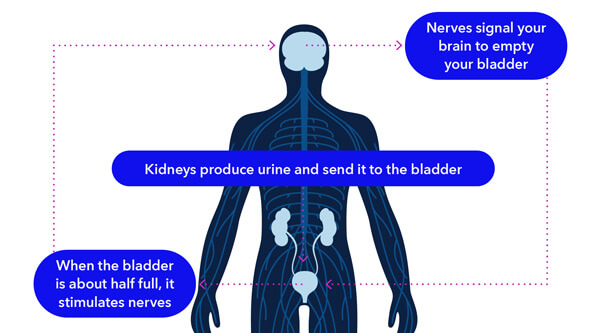
How InterStim™ Works
How the Procedure is Performed
Unlike other bladder control treatments, InterStim™ lets you try it first with an evaluation. The evaluation is performed right in your doctor’s office and involves placing a lead (a thin wire) in the upper part of your buttock. The lead then attaches to a small external device worn discreetly under your clothes.
Most patients can tell within just a few days if it’s working for them, though some evaluations may take up to two weeks. After the evaluation period, you and your doctor will decide if the long-term therapy is right for you. If it is, your evaluation device will be replaced with an implantable device called a neurostimulator during a short outpatient procedure.
Important Considerations
It’s important to remember this therapy is not for everyone. This treatment is prescribed by your doctor. Please talk to your doctor to decide whether this therapy is right for you.
Implanting an InterStim™ system has risks similar to any surgical procedure, including swelling, bruising, bleeding and infection. In addition to risks related to surgery, complications from this therapy can include pain, infection, sensation of electrical shock, device problems, undesirable change in voiding function and lead migration, among others. Talk with your doctor about ways to minimize these risks.
A True Transformation
Of those patients who have found success with InterStim™, many have told us that it has truly transformed their lives. People who had lived for years with leaks and accidents have found they can once again control their bladders, rather than having their bladders control them. No more scheduling activities based on bathroom availability. No more sense that they have to go right now. And no more unpleasant accidents.
If you think InterStim™ might be right for you, our staff is available to discuss your particular condition and treatment options.
Video Overview of Overactive Bladder
The National Association for Continence has developed the video series shown on this page to give you a better sense of what Overactive Bladder is and the treatments available. They’re brief, easy-to-understand presentations that are intended not only to educate you about the condition, but also to help you feel comfortable talking with your physician about your own symptoms.
Medtronic Bladder Control Therapy Delivered by The InterStimTM Systems
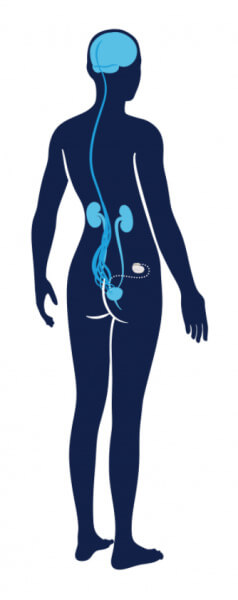
Get more control with the InterStim Systems6,7
- 84% satisfaction among those who use it8
- 3X greater improvements in OAB quality of life9
- 82% of people achieved success† at 5 years6
- Only therapy that lets you see if it works before you and your doctor decide
- More than 375,000 people worldwide have chosen the Medtronic InterStim systems for more control and long-lasting relief6,7
- Recharge-free and rechargeable options let you choose the right device for your lifestyle
In addition to risks related to surgery, complications can include pain at the implant sites, new pain, infection, lead (thin wire) movement/migration, device problems, undesirable changes in urinary or bowel function, and uncomfortable stimulation (sometimes described as a jolting or shocking feeling). Talk with your doctor about ways to minimize these risks.
More Control Starts With Our Smart Programmer
The smart programmer allows you and your healthcare provider to manage your therapy as needed. Unlike previous iterations of our technology that resembled a key fob without a display screen, our Smart Programmer allows you:
Visibility to check your stimulation level on a clear, easy-to-read screen
Adjust your stimulation with confidence that its been changed to the setting you intended
Turn stimulation on or off
Change therapy programs (as directed by your healthcare provider)
Activate MRI mode and check MRI eligibility (see more information below)
Download Brochure


Medtronic Bladder Control Therapy delivered by the InterStim™ system treats urinary retention (inability to completely empty the bladder) and the symptoms of overactive bladder, including urinary urge incontinence (leakage) and significant symptoms of urgency-frequency. It should be used after you have tried other treatments such as medications and behavioral therapy and they have not worked, or you could not tolerate them.
You must demonstrate an appropriate response to the evaluation to be a candidate. You cannot have diathermy (deep heat treatment from electromagnetic energy) if you have an InterStim™ device.
This therapy is not intended for patients with a urinary blockage. Safety and effectiveness have not been established for pregnancy and delivery; patients under the age of 16; or for patients with neurological disease origins.
In addition to risks related to surgery, complications can include pain at the implant sites, new pain, infection, lead (thin wire) movement/migration, device problems, interactions with certain other devices or diagnostic equipment such as MRI, undesirable changes in urinary or bowel function, and uncomfortable stimulation (sometimes described as a jolting or shocking feeling).
This therapy is not for everyone. This treatment is prescribed by your doctor. Please talk to your doctor to decide whether this therapy is right for you. Your doctor should discuss all potential benefits and risks with you. Although many patients may benefit from the use of this treatment, results may vary. For further information, please call Medtronic at 1-800-328-0810 and/or consult Medtronic’s website at www.medtronic.com. USA Rx only. Rev. 0517.

Talking With Your Doctor
For many patients, the hardest part of their treatment plan is getting started. It isn’t always easy to talk about incontinence, even with your physician, but it’s a necessary first step if you want to see results.
Give us a call today at (505) 888-0443 and we’ll help you develop a treatment program
that’s right for you.
Footnotes 1
*Success defined as a 50% or greater reduction in your troublesome bladder symptoms. Numbers reflect completers analysis defined as patients with diary data at baseline and 12 months (n=220). Clinical success was 82% at 12 months using the modified completers analysis (subjects who either had a baseline and 12 month evaluation or withdrew early due to device-related reasons and are considered failures). Device-related adverse events occurred in 30% of subjects post-implant. The most common device-related AEs were unde- sirable change in stimulation, implant site pain, and implant site infection.
† Defined as a 50% or greater reduction in your troublesome bladder symptoms. Numbers reflect completers analysis which is subjects who had evaluable diary data at both baseline and 5 years. Modified Completers analysis was 67% at 5 years. The most common AEs were: undesirable change in stimulation (22%, 60/272); implant site pain (15%, 40/272); and therapeutic product ineffective (13%, 36/272).
References:
1. Leede Research, “Views on OAB: A Study for the National Association of Continence.” December 16, 2015.
2. Noblett K, Siegel S, Mangel J, et al. Results of a Prospective, Multicenter study evaluating quality of life, safety, and efficacy of sacral neuromodulation at twelve months in subjects with symptoms of overactive bladder. Neurourol Urodyn. 2016; 35: 246-251. doi:10.1002/nau.22707
3. Siegel S, Noblett K, Mangel J, et al. Five-Year Follow-up Results of a Prospective, Multicenter Study of Patients with Overactive Bladder Treated with Sacral Neuromodulation. The Journal of Urology 2018;Volume 199(1), 229 – 236.
4. Leng WW, Morrisroe SN. Sacral nerve stimulation for the overactive bladder. Urol Clin N Am. 2006;33:491-501.
5. Chancellor MB, Chartier-Kastler EJ. Principles of sacral nerve stimulation (SNS) for the treatment of bladder and urethral sphincter dysfunctions. Neuromoudlaiton 2000;3(1):15-26.
This web page is made possible by the National Association for Continence.
© 2019, NAFC. All rights reserved.
Footnotes 2
*Restored function defined as a 50% or greater reduction in dysfunctional voiding symptoms from baseline
†Numbers reflect completers analysis defined as patients with diary data at baseline and 12 months (n=220). Clinical success was 82% at 12 months using the modified completers analysis (subjects who either had a baseline and 12-month evaluation or withdrew early due to device-related reasons and are considered failures). Success defined as a 50% or greater reduction in your troublesome bladder symptoms.
‡Under certain conditions; see approved labeling for details. Patients with InterStim™ SureScan™ MRI Leads only.
1. Dasgupta R. Critchley HD, Dolan RJ, Fowler CJ. Changes in brain activity following sacral neuromodulation for urinary retention. J Urol. 2005; 174:2268-2272
2. Griffiths D, Derbyshire S, Stenger A, Resnick N. Brain control of normal and overactive bladder. J Urol. 2005; 174:1862-1867.
3. Griffiths D, Tadic SD. Bladder control, urgency, and urge incontinence: evidence from functional brain imaging. Neurourol Urodyn. 2008;27(6):466-474.
4. Kenefick NJ, Emmanuel A, Nicholls RJ. Effect of sacral nerve stimulation on autonomic nerve function. British Journal of Surgery. 2003;90:1256-1260.
5. Patton V, Wiklendt L, Arkwright JW, Lubowski DZ, Dinning PG. The effect of sacral nerve stimulation on distal colonic motility in patients with fecal incontinence. Br J Surg. 2013;100:959-968.
6. Siegel S, Noblett K, Mangel J, et al. Five-year follow-up results of a prospective, multicenter study of patients with overactive bladder treated with sacral neuromodulation. J Urol. 2018; 199(1), 229-236.
7. Medtronic InterStim Therapy Clinical Summary (2018).
8. Foster RT Sr, Anoia EJ, Webster GD, Amundsen CL. In patients undergoing neuromodulation for intractable urge incontinence a reduction in 24-hr pad weight after the initial test stimulation best predicts long-term patient satisfaction. Neurourol Urodyn. 2007; 26:213-217.
9. Siegel S, Noblett K, Mangel J, et al. Results of a prospective, randomized, multicenter study evaluating sacral neuromodulation with InterStim® Therapy compared to standard medical therapy at 6-months in subjects with mild symptoms of overactive bladder. Neurourol Urodyn. 2015; 34:224-230.
